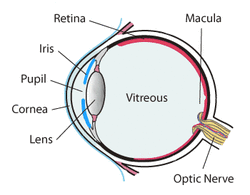
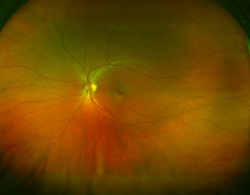
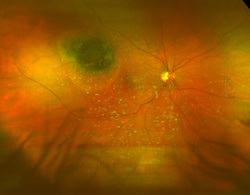
Typically when we go to an optometrist it is with the intention of improving eye sight. But, there is a lot more going on as we will learn. The frequently remembered portion of the eye exam is the test where we look at an eye chart and see how far down the chart that we can read. E, O, M, maybe Z, and in this test called refraction, the optometrist places lenses in front of each eye to determine the best corrective lens strength. This lens correction may be required when the light falling on the retina is either to far or too close due to the eye lens shape. The retina is the area inside of the eye that is like the photo element in a digital camera, or film in older cameras. It contains the rods and cones that allow the light signal to be sent to the optic nerve and provide vision. But, the retina also may contain clues to other problems unrelated to vision. For example, conditions like hypertension, diabetes, melanoma, detached retina and other problems may leave clues that allow the optometrist to suggest further testing leading to early diagnosis of an illness.













 RSS Feed
RSS Feed
